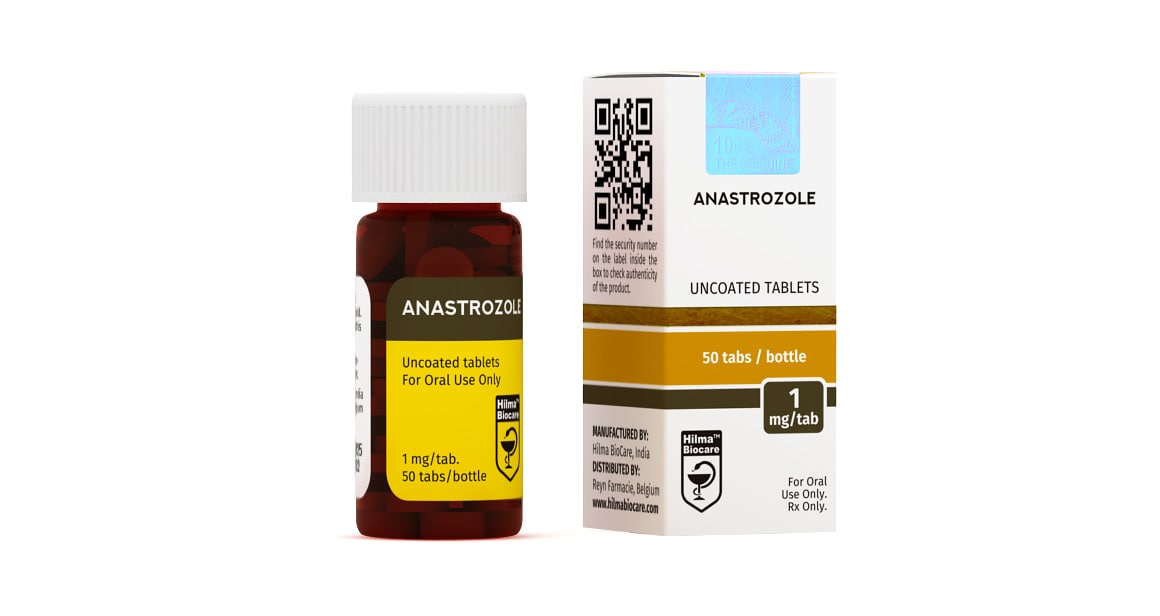
Description
Anastrozole
Strength: 1 mg
Molecular Formula: C17H19N5
Molecular Weight: 293.374 g/mol
Active Ingredient: Anastrozole
CAS number: 120511-73-1
Dosage Form: Tablet
Route: Oral
Market Status: Prescription
Company: Hilma Biocare
DESCRIPTION
Initially Anastrozole was G created in an effort to combat breast cancer in post- menopausal
women. The drug is also used by bodybuilders / powerlifters. Anastrozole is a powerful
anti-estrogen medication, which belongs to the aromatase inhibitor (Al) family. Anastrozole is
not an anabolic steroid, but isole commonly used in conjunction with anabolic steroids to
combat related side-effects.
CLINICAL PHARMACOLOGY
Anastrozole can actively reduce estrogen in the body by as much as 80%. Coupled with its
estrogen reducina effects Anastrozole also greatly increases testosterone in the body and
can do so by as much as 50% by increasing total testosterone as well as Luteinizing
Hormone (LH) and Follicle Stimulating Hormone (FSH) total output.
DIRECTIONS TO USE
Treating certain types of breast cancer in women who have been through menopause,
including women with disease progression after tamoxifen therapy. It may also be used for
male patients with low testosterone levels. Other conditions of treatment should be
determined by your doctor.
ADVERSE REACTIONS
Serious adverse reactions with Anastrozole occurring in less than 1 in 10,000 patients, are:
1) skin reactions such as lesions, ulcers, or blisters; 2) allergic reactions with swelling of the
face, lips, tongue, and/or throat. This may cause difficulty in swallowing and/or breathingB
and 3) changes in blood tests of the liver function, including inflammation of the liver with
symptoms that may include a general feeling of not being well, with or without jaundice, liver
pain or liver swelling. Common adverse reactions (occurring with an incidence of about 10%)
in women taking Anastrozole tablets included: hot flashes, asthenia, arthritis, pain, arthralgia,
hypertension, depression, nausea and vomiting, rash, osteoporosis, fractures, back pain,
insomnia, headache, bone pain, peripheral edema, increased cough, dyspnea, pharyngitis
and lymphedema. Anastrozole may cause dizziness. This effect may be worse if you take it
with alcohol or certain medicines. Use anastrozole with caution. Do not drive or perform
other possibly unsafe tasks until you know how you react to it Anastrozole lowers the
amount of estrogen in your body, which may cause your bones to become softer and
weaker. This may increase your chance of bone fractures. especially of the spine, hip, and
wrist. Discuss any questions or concerns with your doctor.
CONTRAINDICATIONS
Anastrozole tablets are contraindicated in any patient who has shown a hypersensitivity
reaction to the drug or to any of the excipients. Observed reactions include anaphylaxis,
angioedema, and urticaria Anastrozole tablets may cause fetal harm when administered to a
pregnant woman and offer no clinical benefit to premenopausal women with breast cancer.
PRECAUTIONS
Ischemic Cardiovascular Events: in patients with pre-existing ischemic heart disease, an
increased incidence of ischemic cardiovascular events was observed with Anastrozole in the
ATAC trial (17% of patients on Anastrozole and 10% of patients on tamoxifen). Consider risk
and benefits of Anastrozole therapy in patients with pre-existing ischemic heart disease [see
Adverse Reactions. Cholesterol: risk of elevated serum cholesterol compared to patients
receiving tamoxifen (9% versus 3.5%, respectively)
DRUG INTERACTIONS
Warfarin: n a study onem conducted in 16 male volunteers, Anastrozole did not alter the
exposure (as measured by Cmax and AUC) and anticoagulant activity (as measured by
prothrombin time, activated partial thromboplastin time, and thrombin time) of both R- and
S-warfarin.
Cytochrome P450: Based on in vitro and in vivo results, it is unlikely that co- administration
of Anastrozole mg will affect other drugs as a result of inhibition of cytochrome P450
DOSAGE AND ADMINISTRATION
Daily dosage for males is from 0.5mg to 3mg. For women, a maximum dosage of 1mg per
day is enough to combat estrogenic side effects. Because Anastrozole has a short active
life, dosages are usually taken two to six times a day at equal intervals. During PCT, athletes
visiting normally start with higher o dosage then implement and progressive decrease in nem
dosage. The PCT protocol! typically runs for seven to 14 days.
PRESENTATION
Anastrozole 1 mg uncoated tablets: 50 tablets in 1 bottle.
STORAGE
Store between 15 – 25°C. Protecto from light.




Reviews
There are no reviews yet.